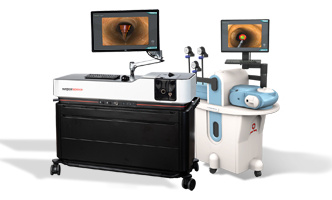
Celebrating 10 Years of Partnership
SAGES and Surgical Science announce their 10-year anniversary of partnership in the…


SAGES and Surgical Science announce their 10-year anniversary of partnership in the…
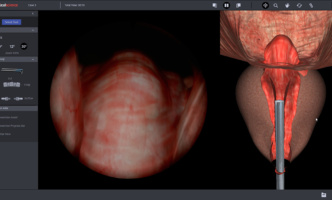
The new modules allow urology trainees to practice treatment of BPH in a safe environment.

How data-enhanced simulation training is driving measurable success in healthcare…

Young minds get a glimpse into medical learning with advanced technology.

Dr. Steinhard emphasized the effectiveness of simulator-based training, particularly…

Surgical Science celebrates the holiday spirit with sponsorship of Team Rynkeby,…
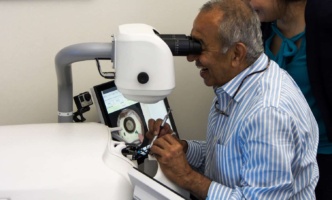
Eliminating cataract blindness through surgical simulation training.

Trento University's Cutting-Edge Lab Integrates Technology and Medicine for Enhanced…

Surgical Science Helps Shape the Future of Human-Machine Interaction. "We are excited…
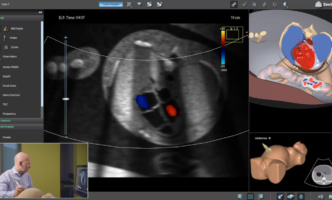
Mastering fetal echo training goes hand in hand with understanding the intricacies…

More than half the world’s population has no access to basic IR procedures.

Robotic surgery is growing in India, catering to the largest population in the world.